iSMIT
The international Society for Medical Innovation and Technology
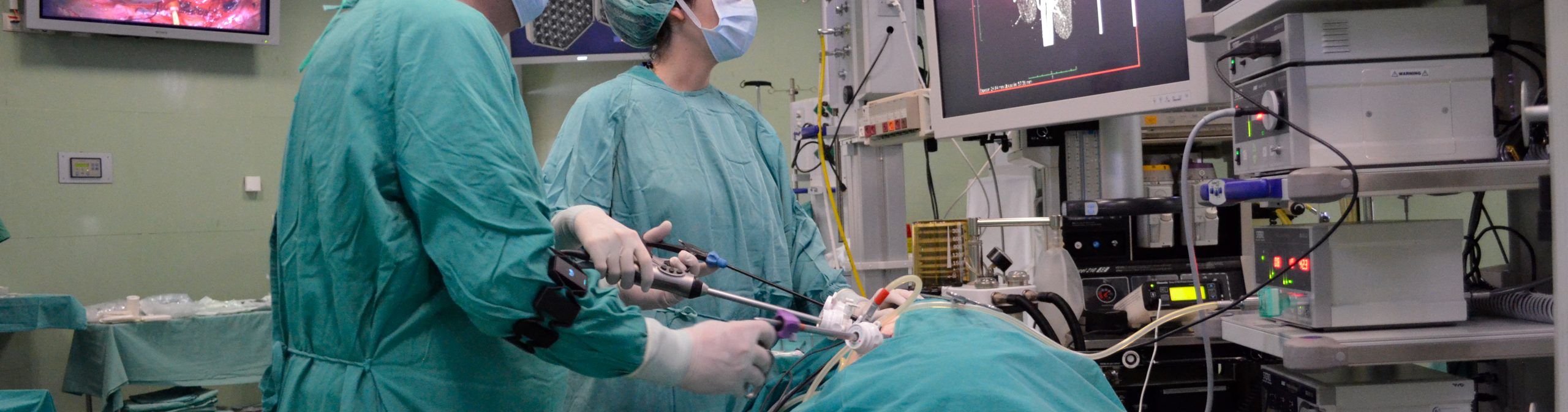
We are an international society, formed in 1989 under the name “Society for Minimally Invasive Therapy” (SMIT) by an innovative group of medical practitioners led by John Wickham. We were dedicated to the multidisciplinary advancement of minimally invasive therapy, which is intended to reduce the patient trauma after traditional surgical or radiological interventions.
Upcoming SMIT conferences
-
35th Annual SMIT Conference
September 26-28, 2024 in Cáceres, SpainConference President:
Francisco M. Sánchez-Margallo, MDCo President:
Juan A. Sánchez-Margallo, MD -


36th Annual SMIT Conference
October 9-11, 2025 in Washington, USAConference President:
Kevin Cleary, PhDCo President:
Anthony Sandler, MD -


37th Annual SMIT Conference
2026 in Munich, GermanyConference President:
Konrad Karcz, MD -


38th Annual SMIT Conference
2027 in Osaka, JapanConference President:
Kiyokazu Nakajima, MD, PhD, FACS -


39th Annual SMIT Conference,
2028 in Rotterdam, The NetherlandsConference President:
John J. van den Dobbelsteen, Prof. Dr.